|
R (hclust, v3.2.4) was used for variant frequency clustering analysis to show what types of samples from cancer patients are more similar. Statistical analysis: For variant frequency less than 0.5%, 0 was replaced. Software ANNOVAR (v) and snpEff (v4.2) was used for variant annotation Program Samtools and VarScan (v2.4.1) was used for variant calling: (1) the average total coverage depth was defined as >1000 and each variant coverage as >10 for called variant, at least one sample with variant frequency >1%, variant frequency of each sample >0.5%, and P value <0.01 (2) visual examination of the mutations was performed using Samtools software () and possible errors specific to one DNA strand were filtered out. Raw reads were mapped to reference genome hg19 by using BWA (0.7.12-r1039). 
Variant calling: Initial data from HiSeq X-Ten were evaluated by using fastQC (v0.11.3). The AmpliSeq Cancer Panel covers 1406 cancer-associated genes which developed by Co. 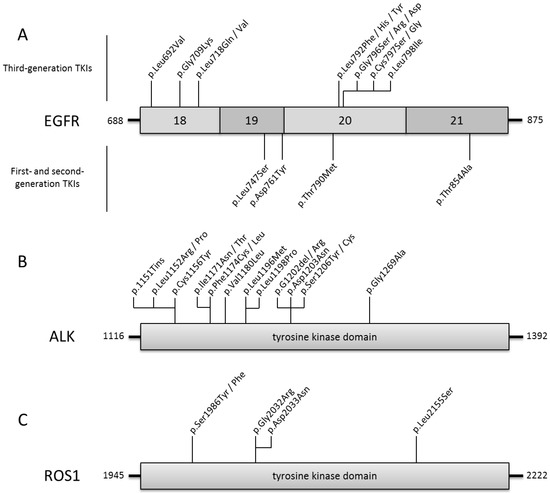
All samples were subjected to Illumina HiSeq X-Ten for paired-end sequencing (150bp each end). Targeted amplification and Illumina adapter-ligated library preparation was performed using Amplicon Sequencing-Illumina Compatible Kit following manufacturer's instructions (Questgenomics, Nanjing, PRC). 
Library preparation and sequencing: For each sample, DNA was quantified with the Qubit dsDNA HS Assay kit (Life Technologies,USA) as manufacturer's recommended protocol. All kits were used according to the manufacturers' instructions. QIAamp Circulating Nucleic Acid Kit (Qiagen, German) was used to extract cfDNA form plasma. The supernatants were centrifuged again at 10,000 g for 10 min, and plasma was collected and stored at -80☌.Tiangen whole blood DNA Kit (Tiangen, Beijing, PRC) were used to extracted DNA from peripheral blood lymphocytes, respectively. The cell pellets containing peripheral blood lymphocytes were stored at -20 ☌. 10mL tubes containing blood samples with EDTA added were centrifuged at 1000g for 10min. Sample DNA handling: Peripheral blood lymphocytes (PBLs), and plasma were collected for analysis for each patient. Why Should I Register and Submit Results?.She has been recipient of two research grants (2015, My First AIRC Grant for the translational project “MET-targeting in Small Cell Lung Cancer 2010, ESMO translational research fellowship award). Pasello is extensively published in peer-review journals with 75 papers (H index: 16). Main research fields are: tumor immune microenvironment and circulating biomarkers of inflammation in thoracic cancers and real world studies on oncology diagnostic-therapeutic pathways and innovative drugs access. To date, she has been the Principal Investigator of more than 30 clinical trials and translational research projects, with particular reference to new treatment targets discovery and innovative drugs development in thoracic cancers. Pasello is the reference medical oncologist for thoracic cancer at the Istituto Oncologico Veneto, a research and care institute. 
Giulia Pasello (MD PhD) is Assistant Professor (Ricercatore a tempo determinato B) at the Department of Surgery, Oncology and Gastroenterology, University of Padova. In order to guarantee equity in treatment, these considerations should find their application widely and rapidly.Īim of this review is offering an overview of emerging biomarkers, relative upcoming targeted drugs, and new diagnostic chances with an authors’ perspective about a real-life diagnostic-therapeutic algorithm useful for daily clinical practice. Genomic data should be than integrated in the clinical context and matched with available therapeutic options Molecular Tumor Boards (MTB) are worldwide emerging interdisciplinary groups implemented to transfer the impact of precision medicine in clinical practice. Crucial preliminary step is the selection of the best available sample before testing, aware of clinical condition and setting of disease. Nevertheless, several aspects impact on success rate of NGS in clinical practice: a multidisciplinary approach and thorough knowledge of strengths and limits of each technologic diagnostic tool are required. The increasing number of approved drugs along with next generation sequencing (NGS) technologies look out as potential revolution of biomolecular characterization of non-small-cell lung cancer (NSCLC).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
